
Join the U2 MB Knee
Follow-Up Study
Take part in a UK 10-year clinical follow-up study of the U2 Mobile Bearing (MB™) Knee System. We collect questionnaires, routine X-rays, and long-term outcome data.

Study sponsor: United Orthopedic Inc.
Who Can Join
This Study
Who are we looking for?
Patients who have had, or are scheduled to have, a total knee replacement using the U2 Mobile Bearing (MB™) System.
Study Protocol: This is a prospective follow-up study of an approved implant. It is not an interventional treatment study.
Functional Performance
We track knee function and day-to-day mobility over 10 years.
Patient-Reported Outcomes
We collect validated patient-reported outcomes on pain, function, and quality of life.
Implant Integrity
Routine follow-up data helps monitor implant positioning, stability, and survivorship.
Why It Matters
The study adds UK long-term evidence to support future care decisions.
Interested in taking part?
Bridging the Gap in
Knee Arthroplasty
Total knee replacement is a cornerstone of modern orthopaedics, offering relief for end-stage osteoarthritis. This study looks beyond revision rates alone.
The Unmet Need
Some patients still report pain, stiffness, or functional limits after surgery.

U2 MB™ Technology
The mobile-bearing design is intended to support natural knee movement and long-term function.
UK Specific Evidence
Long-term UK follow-up data is limited. This study adds evidence from routine British clinical practice.
Study Objectives
PROMs
Validated patient-reported outcome measures focusing on daily function.
Radiographic Analysis
Routine structured X-rays to monitor implant positioning and stability.
Safety & Longevity
Long-term tracking of survivorship and post-operative safety data.
Evidence Base
Building a longitudinal database to inform future clinical decision-making.
Interested in taking part?
Your Journey in Research
The study is designed to fit around routine follow-up care as much as possible.
Questionnaires
You will complete validated knee questionnaires about pain, function, and satisfaction.
Routine X-rays
AP and lateral knee X-rays are collected at follow-up visits to assess alignment and implant performance.
Clinical Data
With your consent, the research team records relevant clinical information securely for study analysis.
Interested in taking part?
Study Journey (10 Years)
A structured follow-up schedule to track recovery and implant performance over time.
Screening / Baseline
- Informed consent and eligibility verification.
- Collection of basic clinical details and initial questionnaires.
Follow-up Milestones
Knee function & satisfaction questionnaires.
Routine standard X-rays (AP & Lateral views).
Progress Review
Review of clinical events, knee function, and patient-reported outcomes.
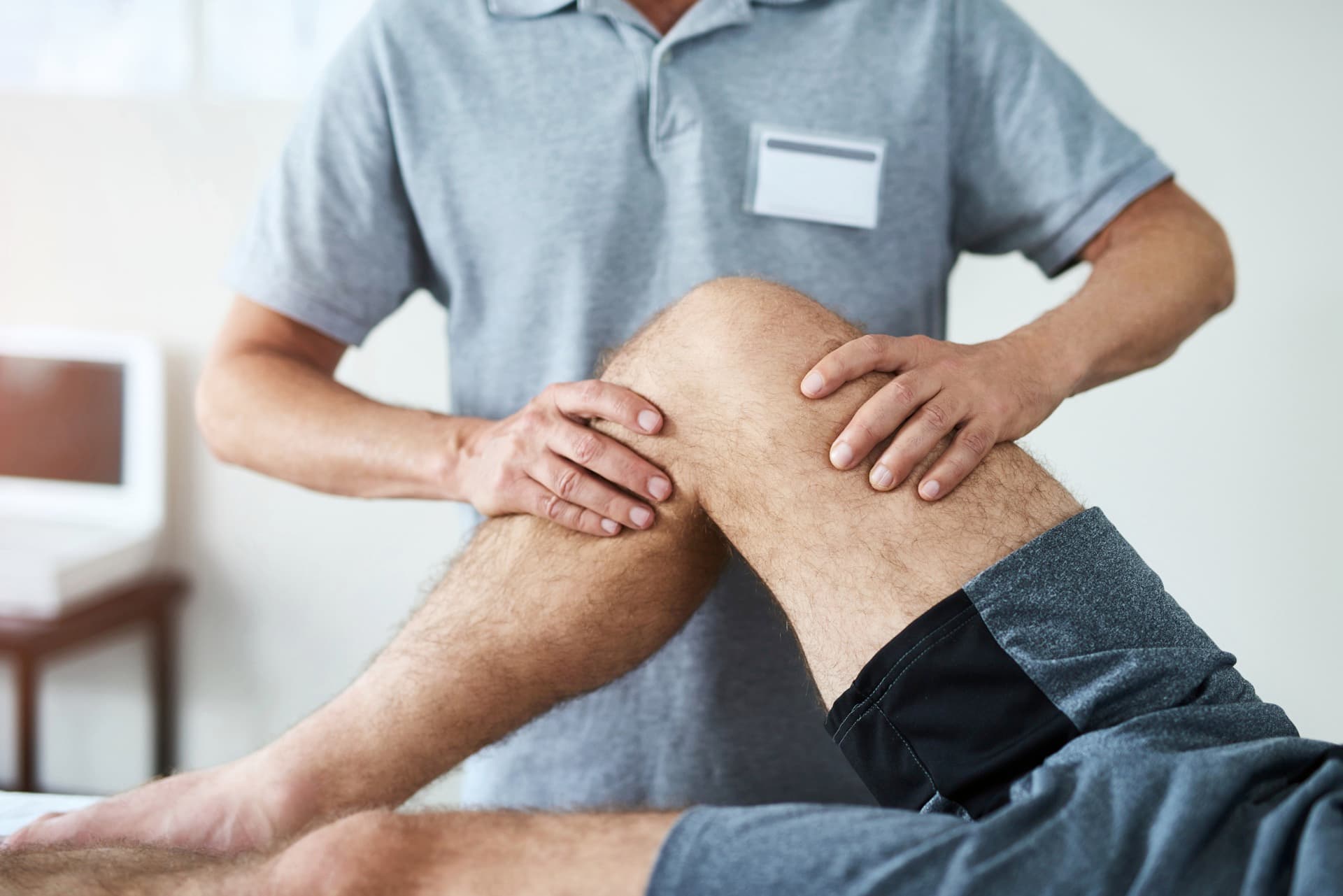
Final Long-Term Review
Final review of implant survivorship and quality-of-life outcomes.
Note: These visits are carefully scheduled to align with standard NHS or private follow-up pathways. In most cases, the study adds questionnaires and structured data collection to routine appointments.
Interested in taking part?
Participation Criteria
Please review the key eligibility requirements before registering your interest.
Who Can Take Part
General Inclusion Requirements
Age Requirement
Participants must be aged 21 years or over at the time of recruitment.
Clinical Pathway
Scheduled for or have received a primary total knee replacement (TKR) specifically using the U2 MB Knee System.
Commitment to Care
Ability to provide informed consent and a willingness to attend the 10-year follow-up schedule.
Exclusion Criteria
- Active joint infections or clinical contraindications.
- Conditions such as malignancy or neuromuscular disorders that may preclude follow-up.
- Inability or unwillingness to comply with long-term data collection.

Our research team is available to discuss your eligibility in detail during your clinic visit.
Why Take Part
Your treatment does not change, but your follow-up data can help improve understanding of long-term outcomes with the U2 MB Knee System.
Advancing Surgical Excellence
Add to the evidence used to assess long-term outcomes after knee replacement.
Empowering Future Patients
Help future patients and clinicians make better-informed decisions.
Defining Functional Longevity
Contribute data on knee function, satisfaction, and survivorship over a decade.

Risk Assessment & Transparency
This is a non-interventional follow-up study. The main considerations are time, questionnaires, and routine imaging.
Participation Effort
Minor discomfort or time commitment required while completing detailed functional and health questionnaires.
Routine Imaging
Exposure to a minimal amount of ionising radiation from routine knee X-rays, which are already part of your standard clinical care.
Surgical Context
Standard risks of knee replacement (infection, loosening) are associated with the surgery itself, not the research procedures.
Non-Interventional Guarantee
Research participation does not involve any experimental drugs, injections, or invasive treatments.
Interested in taking part?
Ethics, Governance & Oversight
The study is run under formal ethics, governance, and data protection standards.
Approving Body
Health Research Authority (HRA)
REC 25/WM/0048
Data Protection
Managed in line with UK GDPR and the Data Protection Act.
GCP Compliance
Managed in accordance with Good Clinical Practice (GCP) guidelines.
Sponsorship
Sponsored by United Orthopedic Inc. in partnership with MSK Doctors.
Interested in taking part?
Research Leadership
Sponsor and clinical partner for this study.
United Orthopedic Inc.
Sponsor of the study and manufacturer of the U2 MB Knee System.

MSK Doctors
Clinical partner responsible for patient oversight and study data collection.
